We accelerate organ model research
Philip Ezze, CC BY-SA 4.0, via Wikimedia Commons
Perfusion system for vascularized, printed organ models
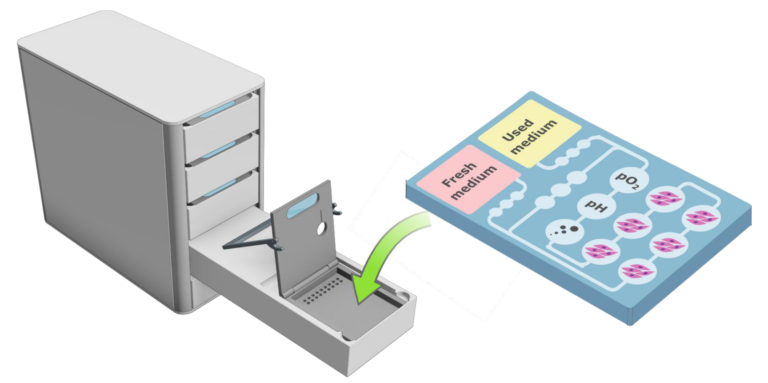
Setup of a multi-process bioreactor for vascularized tissue models
Bioprinting has enjoyed rapidly growing popularity in recent years. However, the vast majority of printed tissue models fall far short of the theoretical capabilities of the technology due to the lack of vascular structures and also the lack of appropriate culture systems to produce such high-quality tissue models. We aim to remove this limitation and massively expand the application scope of bioprinting and significantly increase the quality of 3D-printed tissue models by developing a novel automated culturing system for vascularized tissue models.
Bioprinting provides spatial coarse structures that subsequently lead to the formation of physiological tissue structures through self-organization processes. For example, endothelial cells form microcapillaries in the tissue during long-term dynamic cultivation, and cells arrange themselves into clusters and form cell-cell interactions. It is only through this long-term cultivation under dynamic conditions that the printed gross structures become physiologically relevant biological tissues.
Co-financed by the European Regional Development Fund
